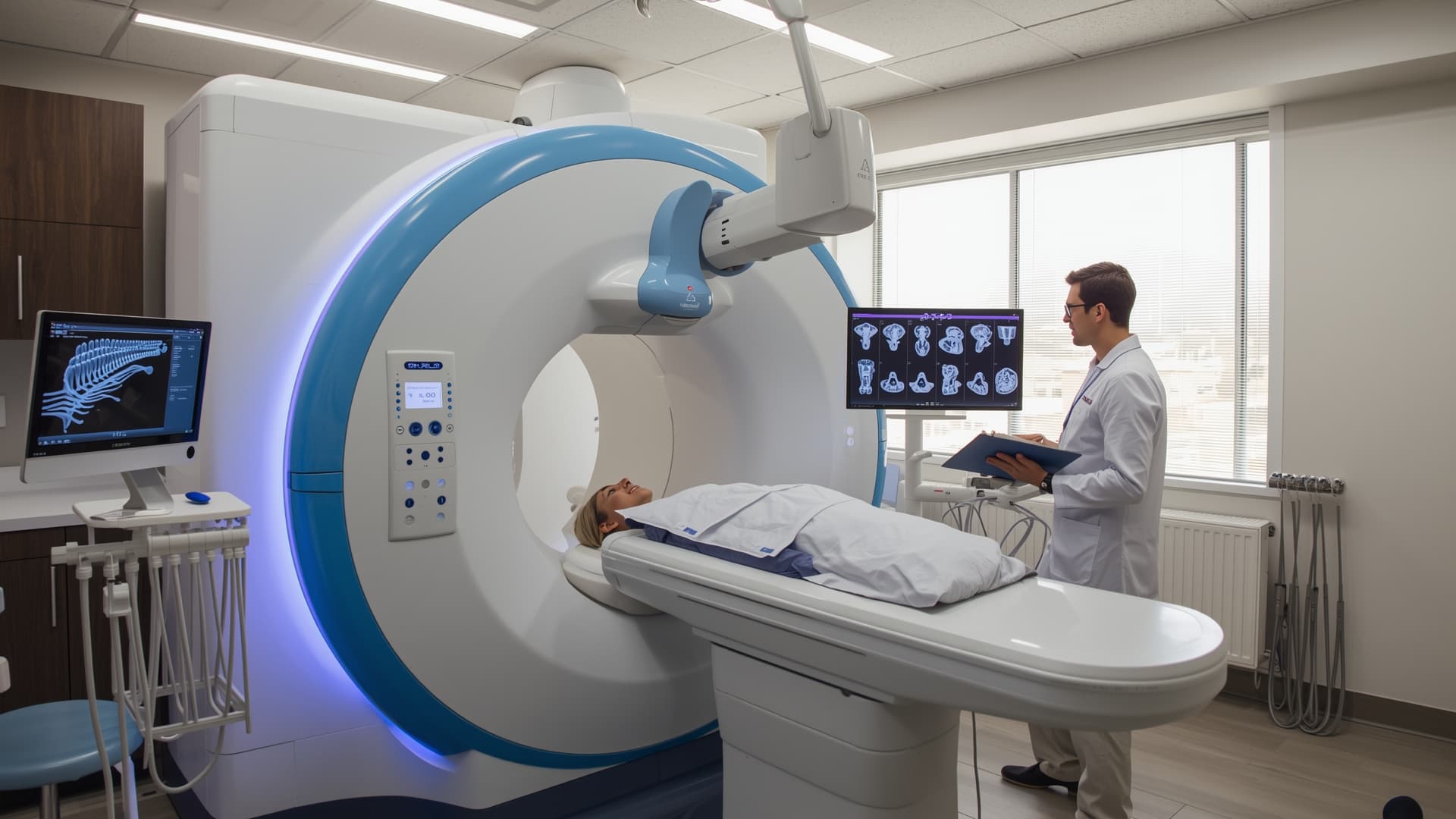
For decades, dental imaging meant one thing: radiation. Intraoral X-rays, panoramic films, and CBCT scans all rely on ionizing radiation to produce images. That changed in March 2026 when the FDA cleared the first dental-dedicated MRI system, opening a new chapter in how clinicians visualize soft tissue without exposing patients to a single X-ray.
What Happened
Dentsply Sirona and Siemens Healthineers announced on March 9, 2026, that their jointly developed MAGNETOM Free.Max Dental Edition received FDA clearance in the United States. The system is the first MRI built specifically for dentomaxillofacial diagnostics.
The clearance followed a clinical trial that validated the system across five dental specialties: endodontics, periodontics, TMJ assessment, tooth extraction planning, and orthodontics. Key clinical findings from the trial include:
- Inflammation detection: The system can differentiate active inflammation from healthy tissue and scar tissue.
- Pulp vitality assessment: Non-invasive imaging of tooth pulp to complement chairside vitality testing.
- Tooth-nerve visualization: Imaging of teeth and their spatial relationship to neighboring teeth and nearby nerves.
The device uses a dedicated dental coil and specialized software to focus on dentomaxillofacial structures without imaging brain tissue. Average workflow time, including patient setup, comes in under 20 minutes.
Why It Matters for Your Practice
The dental imaging market is projected to exceed $4 billion by 2030, and AI-driven diagnostic tools are already reshaping how practices interpret scans. Dental-dedicated MRI adds an entirely new imaging modality to the mix, and it addresses a real clinical gap.
Soft-tissue contrast without radiation. CBCT gives you excellent bone and hard-tissue detail, but soft-tissue visualization has always been its limitation. MRI excels exactly where CBCT has natural limitations. For endodontic cases where you need to assess pulp health, or periodontal cases where you need to distinguish active inflammation from fibrosis, dental MRI provides information that was previously difficult to obtain.
Complementary, not a replacement. Dentsply Sirona and Siemens Healthineers have been clear: this technology is designed to work alongside intraoral, panoramic, and CBCT imaging. It fills diagnostic gaps in cases where soft-tissue characterization is critical. Your existing imaging workflow does not become obsolete.
Initial target audience is institutional. The MAGNETOM Free.Max Dental Edition is primarily intended for hospitals, large dental clinics, and universities. If you run a solo or small group practice, this is not something you will be purchasing next quarter. But referral pathways to institutions with dental MRI capability could soon become part of your treatment-planning toolkit, similar to how practices currently refer out for medical CT or MRI when needed.
Training is already underway. Dentsply Sirona showcased the system at the ADEA Annual Session in Montreal (March 21-24, 2026), and a hands-on clinician training program launched in April 2026 at the University of Minnesota covering imaging protocols, clinical procedures, and image interpretation.
The Radiation Conversation Is Shifting
The dental profession has worked hard to minimize radiation exposure through digital sensors, collimation, and ALARA principles. CBCT, while valuable, still delivers meaningfully higher doses than periapical films. The FDA and ADA both recommend limiting CBCT to cases where lower-dose modalities cannot answer the clinical question.
Dental MRI removes radiation from the equation entirely for soft-tissue assessment. For pediatric patients, pregnant patients, or any case requiring repeat imaging over time, that is a meaningful clinical advantage. As imaging technology continues to evolve, expect radiation-free modalities to gain ground.
TMR Take: This is a genuine milestone, not a marketing exercise. A radiation-free imaging modality purpose-built for dental diagnostics has been a research goal for years, and the clinical trial data backing this clearance is substantive. That said, the near-term impact on most private practices will be indirect. Watch for referral networks to develop around institutional early adopters, and keep an eye on how the technology evolves toward smaller-footprint, practice-friendly configurations over the next two to three years.
What to Do Now
- No immediate action required. Your current imaging protocols remain the standard of care. Dental MRI is an addition to the diagnostic toolkit, not a disruption to existing workflows.
- Follow the referral pathway. If you are near a university dental center or large hospital system, ask whether they plan to adopt dental MRI. Knowing where to refer complex soft-tissue cases will set you apart.
- Stay current on training. Dentsply Sirona has announced clinician training programs. Even if you will not operate the equipment, understanding how to read and interpret dental MRI findings will be valuable as the technology reaches more institutions.
- Brief your team. Patients will see headlines about radiation-free dental imaging. Have a clear, honest answer ready: this is real, it is complementary to X-rays and CBCT, and it is currently available at select institutions.
The Bottom Line
The FDA clearance of the MAGNETOM Free.Max Dental Edition marks the arrival of a new imaging modality in dentistry. For now, the technology lives in hospitals and universities. Over time, as the platform matures and becomes more accessible, dental MRI could meaningfully change how clinicians approach soft-tissue diagnostics. The practices that stay informed now will be best positioned when that shift arrives.
Want to keep up with the technology shaping modern dental practices? Browse our dental AI coverage or take our software-match quiz to find the right tools for your practice.